Deep Brain Stimulation Offers New Hope for Severe Depression Patients
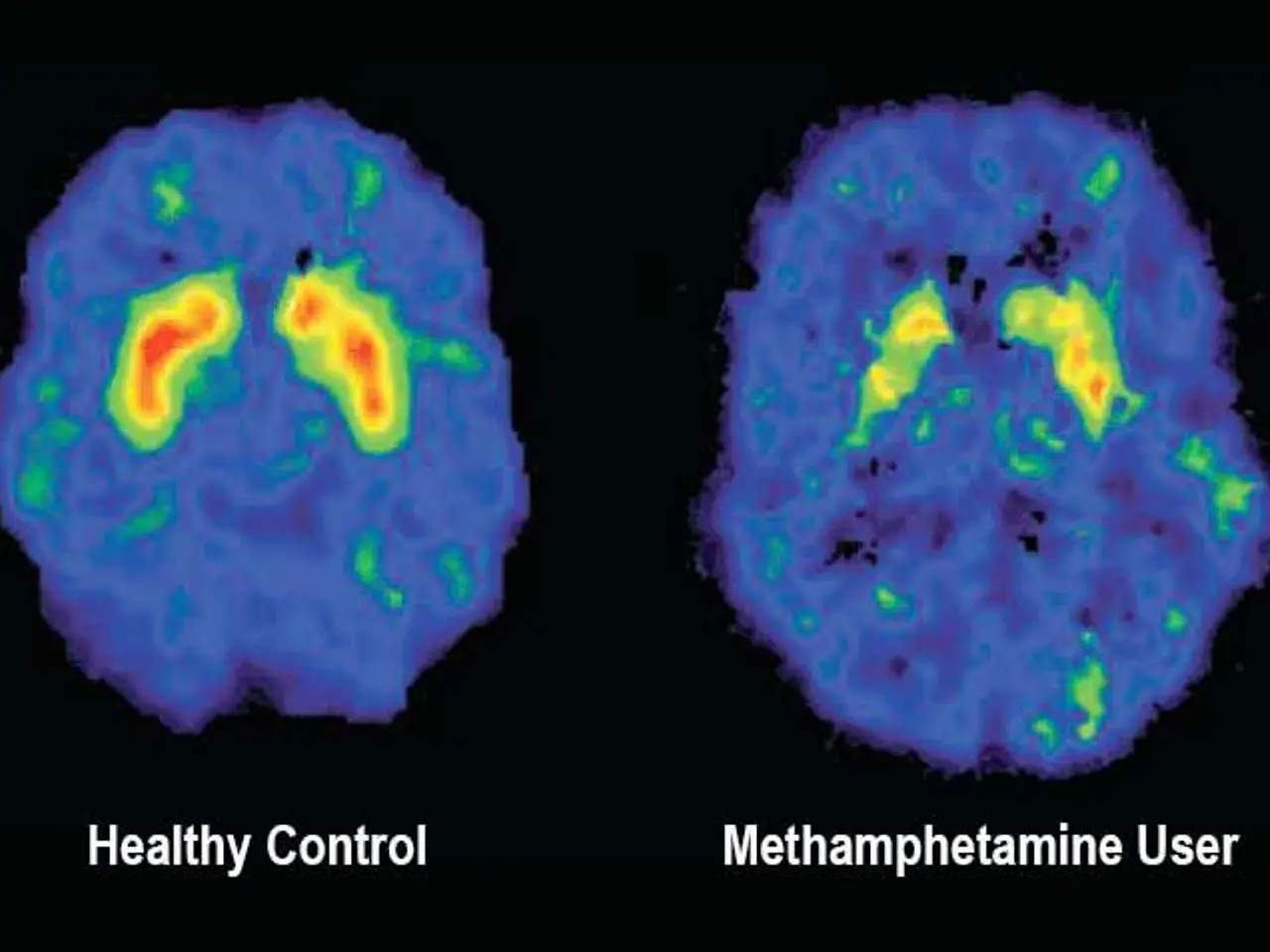
A new approach to treating severe depression is being tested at UT Southwestern (UTSW) Medical Center. Known as deep brain stimulation (DBS), the therapy targets patients who have found no relief from existing treatments. Early results suggest it could offer hope where other methods have failed. DBS is already approved by the FDA for conditions like Parkinson's disease, where it acts like a 'pacemaker for the brain'. Electrode implants deliver controlled electrical pulses to specific brain areas. For Parkinson's, these pulses target grey matter, often producing rapid improvements in movement.
For depression, the approach differs. Instead of grey matter, DBS focuses on the brain's 'white matter'—the communication pathways that connect different regions. The goal is to 'unstick' a brain trapped in a chronic depressive state, allowing it to function more flexibly. Unlike the quick effects seen in Parkinson's, changes from DBS for depression develop slowly, sometimes taking weeks or months.
UTSW is one of the sites for the TRANSCEND trial, a rigorous double-blind study assessing DBS for treatment-resistant depression. Candidates for the trial include adults who have struggled with severe, long-term depression and failed to respond to at least four other treatments. Around 30% of people with depression fall into this category, leaving them with few options.
Preliminary findings indicate DBS may help reduce biological barriers that keep the brain locked in depression. This could enable patients to re-engage in daily activities and therapies that previously seemed out of reach. However, the treatment remains experimental and is only available through clinical trials, as it has not yet received FDA approval for depression. The TRANSCEND trial continues to explore whether DBS can provide lasting relief for those with treatment-resistant depression. While early results are promising, the therapy is still under investigation and not yet widely accessible. Patients interested in participating must meet strict criteria and enrol through approved study sites like UTSW.