FDA Debate Over SSRIs in Pregnancy Sparks Fears of Overwarning
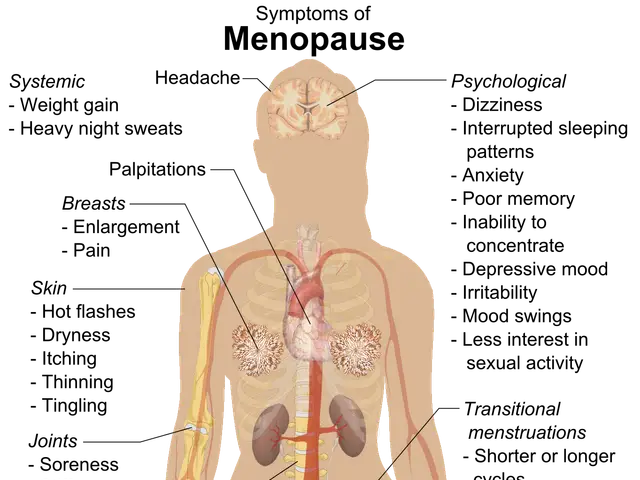
A debate over antidepressant safety during pregnancy has intensified after a leading FDA official accelerated the review of a petition calling for stricter warnings. Dr. Adam Urato, a maternal-fetal medicine specialist, wants the agency to add a boxed warning to SSRIs—linking them to miscarriages, autism, and fetal brain abnormalities. The move has sparked concern among psychiatrists, who warn that such labels could push women to stop treatment, risking severe consequences from untreated depression.
Over the past decade, public and medical views on SSRIs in pregnancy have shifted. Early fears, fuelled by 2011 FDA alerts and media reports, highlighted possible birth defects and neonatal complications. But later research, including major studies in JAMA Psychiatry and The Lancet, found only modest risks—such as a 20–30% higher chance of cardiac malformations and temporary breathing issues in newborns. Medical groups like ACOG and the APA now recommend tailored assessments, favouring sertraline or citalopram for moderate-to-severe depression while suggesting therapy for milder cases.
Dr. Urato's petition argues that current warnings understate risks like autism and developmental disorders. His push gained urgency when Dr. Tracy Beth Hoeg, the FDA's top drug regulator, intervened to speed up the review. Critics note her close ties to Urato, raising questions about impartiality. The FDA must legally respond to all citizen petitions, but Hoeg's direct involvement has shortened the usual timeline.
Outside experts warn that a new warning could backfire. Dr. Jennifer Payne, a reproductive psychiatrist, argues the petition overlooks the dangers of untreated depression—including self-harm, substance misuse, and poor pregnancy outcomes. With over 15% of US women on antidepressants, doctors already weigh medication risks against relapse harms. Previous label updates, like the 2004 black-box warning on youth suicide risk, show how such changes can reshape treatment decisions.
The FDA's decision will determine whether SSRIs carry a stronger pregnancy warning. If approved, the change could alter how millions of women manage depression during pregnancy. But psychiatrists stress that abrupt medication stops pose their own dangers—leaving patients and doctors to navigate conflicting advice.