Psilocybin therapy shows promise for depression, PTSD, and anxiety in USF trials
Researchers at the University of South Florida (USF) are studying psilocybin, the compound found in 'magic mushrooms', as a potential treatment for mental health disorders. Conditions like anxiety, depression, and PTSD could benefit from this approach. The work builds on earlier clinical trials showing promising results for medically supervised use.
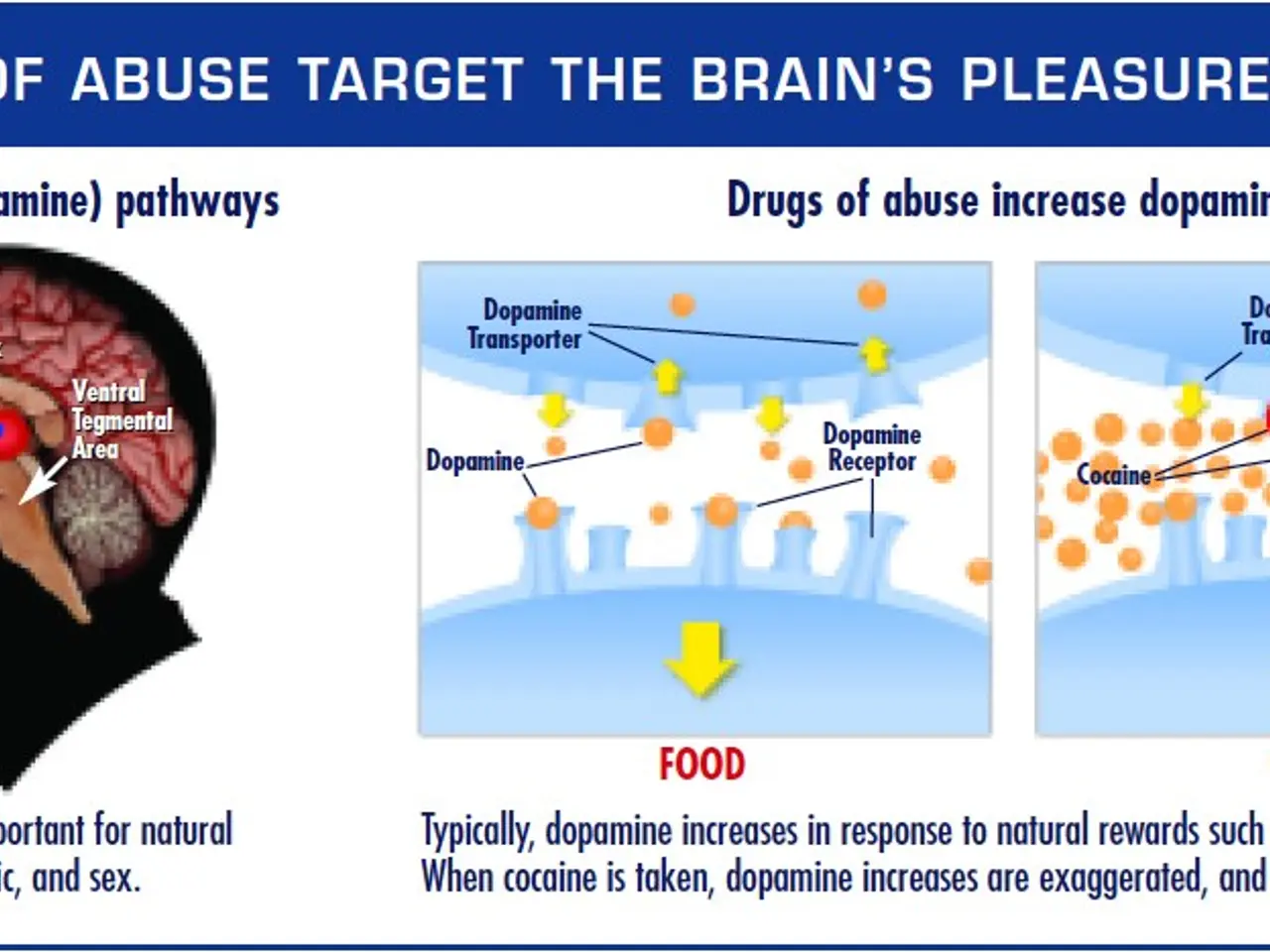
Psilocybin works by targeting serotonin receptors in the brain, according to Dr. Wagoner, who leads USF's Psychiatry & Behavioral Neurosciences Department. He suggests combining the drug with psychotherapy to achieve the best outcomes for patients.
The US trials aim to gather enough evidence to eventually sell the treatment to a private company for FDA approval. This follows broader shifts in the agency's stance on psychedelics. Once cautious, the FDA now includes a dedicated chapter on psychedelic research in its guidelines. Political figures like Health Secretary Robert F. Kennedy Jr. and FDA head Dr. Marty Makary have shown support, though some agency staff remain hesitant.
A recent milestone came on 17 February, when Compass Pathways released positive Phase-III data for psilocybin in treatment-resistant depression. Their approval application is now expected soon. While USF's specific trial details are not yet public, more information can be found on the USF Health website.
If successful, the combination of psilocybin and psychotherapy could offer a new path for treating stubborn mental health conditions. The next steps depend on regulatory reviews and further research. The outcome may influence how similar therapies are developed and approved in the future.